
 Play media
Play media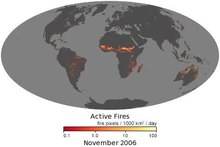 Play media
Play mediaFire is the rapid oxidation of a material in the exothermic chemical process of combustion, releasing heat, light, and various reaction products. Slower oxidative processes like rusting or digestion are not included by this definition.
Fire is hot because the conversion of the weak double bond in molecular oxygen, O2, to the stronger bonds in the combustion products carbon dioxide and water releases energy (418 kJ per 32 g of O2); the bond energies of the fuel play only a minor role here. At a certain point in the combustion reaction, called the ignition point, flames are produced. The flame is the visible portion of the fire. Flames consist primarily of carbon dioxide, water vapor, oxygen and nitrogen. If hot enough, the gases may become ionized to produce plasma. Depending on the substances alight, and any impurities outside, the color of the flame and the fire's intensity will be different.
Fire in its most common form can result in conflagration, which has the potential to cause physical damage through burning. Fire is an important process that affects ecological systems around the globe. The positive effects of fire include stimulating growth and maintaining various ecological systems.
The negative effects of fire include hazard to life and property, atmospheric pollution, and water contamination. If fire removes protective vegetation, heavy rainfall may lead to an increase in soil erosion by water. Also, when vegetation is burned, the nitrogen it contains is released into the atmosphere, unlike elements such as potassium and phosphorus which remain in the ash and are quickly recycled into the soil. This loss of nitrogen caused by a fire produces a long-term reduction in the fertility of the soil, which only slowly recovers as nitrogen is "fixed" from the atmosphere by lightning and by leguminous plants such as clover.
Fire has been used by humans in rituals, in agriculture for clearing land, for cooking, generating heat and light, for signaling, propulsion purposes, smelting, forging, incineration of waste, cremation, and as a weapon or mode of destruction.
Contents
Physical properties
Chemistry
Fires start when a flammable or a combustible material, in combination with a sufficient quantity of an oxidizer such as oxygen gas or another oxygen-rich compound (though non-oxygen oxidizers exist), is exposed to a source of heat or ambient temperature above the flash point for the fuel/oxidizer mix, and is able to sustain a rate of rapid oxidation that produces a chain reaction. This is commonly called the fire tetrahedron. Fire cannot exist without all of these elements in place and in the right proportions. For example, a flammable liquid will start burning only if the fuel and oxygen are in the right proportions. Some fuel-oxygen mixes may require a catalyst, a substance that is not consumed, when added, in any chemical reaction during combustion, but which enables the reactants to combust more readily.
Once ignited, a chain reaction must take place whereby fires can sustain their own heat by the further release of heat energy in the process of combustion and may propagate, provided there is a continuous supply of an oxidizer and fuel.
If the oxidizer is oxygen from the surrounding air, the presence of a force of gravity, or of some similar force caused by acceleration, is necessary to produce convection, which removes combustion products and brings a supply of oxygen to the fire. Without gravity, a fire rapidly surrounds itself with its own combustion products and non-oxidizing gases from the air, which exclude oxygen and extinguish the fire. Because of this, the risk of fire in a spacecraft is small when it is coasting in inertial flight.This does not apply if oxygen is supplied to the fire by some process other than thermal convection.
Fire can be extinguished by removing any one of the elements of the fire tetrahedron. Consider a natural gas flame, such as from a stove-top burner. The fire can be extinguished by any of the following:
- turning off the gas supply, which removes the fuel source;
- covering the flame completely, which smothers the flame as the combustion both uses the available oxidizer (the oxygen in the air) and displaces it from the area around the flame with CO2;
- application of water, which removes heat from the fire faster than the fire can produce it (similarly, blowing hard on a flame will displace the heat of the currently burning gas from its fuel source, to the same end), or
- application of a retardant chemical such as Halon to the flame, which retards the chemical reaction itself until the rate of combustion is too slow to maintain the chain reaction.
In contrast, fire is intensified by increasing the overall rate of combustion. Methods to do this include balancing the input of fuel and oxidizer to stoichiometric proportions, increasing fuel and oxidizer input in this balanced mix, increasing the ambient temperature so the fire's own heat is better able to sustain combustion, or providing a catalyst; a non-reactant medium in which the fuel and oxidizer can more readily react.
Flame
A flame is a mixture of reacting gases and solids emitting visible, infrared, and sometimes ultraviolet light, the frequency spectrum of which depends on the chemical composition of the burning material and intermediate reaction products. In many cases, such as the burning of organic matter, for example wood, or the incomplete combustion of gas, incandescent solid particles called soot produce the familiar red-orange glow of 'fire'. This light has a continuous spectrum. Complete combustion of gas has a dim blue color due to the emission of single-wavelength radiation from various electron transitions in the excited molecules formed in the flame. Usually oxygen is involved, but hydrogen burning in chlorine also produces a flame, producing hydrogen chloride (HCl). Other possible combinations producing flames, amongst many, are fluorine and hydrogen, and hydrazine and nitrogen tetroxide. Hydrogen and hydrazine/UDMH flames are similarly pale blue, while burning boron and its compounds, evaluated in mid-20th century as a high energy fuel for jet and rocket engines, emits intense green flame, leading to its informal nickname of "Green Dragon".
The glow of a flame is complex. Black-body radiation is emitted from soot, gas, and fuel particles, though the soot particles are too small to behave like perfect blackbodies. There is also photon emission by de-excited atoms and molecules in the gases. Much of the radiation is emitted in the visible and infrared bands. The color depends on temperature for the black-body radiation, and on chemical makeup for the emission spectra. The dominant color in a flame changes with temperature. The photo of the forest fire in Canada is an excellent example of this variation. Near the ground, where most burning is occurring, the fire is white, the hottest color possible for organic material in general, or yellow. Above the yellow region, the color changes to orange, which is cooler, then red, which is cooler still. Above the red region, combustion no longer occurs, and the uncombusted carbon particles are visible as black smoke.
The common distribution of a flame under normal gravity conditions depends on convection, as soot tends to rise to the top of a general flame, as in a candle in normal gravity conditions, making it yellow. In micro gravity or zero gravity, such as an environment in outer space, convection no longer occurs, and the flame becomes spherical, with a tendency to become more blue and more efficient (although it may go out if not moved steadily, as the CO2 from combustion does not disperse as readily in micro gravity, and tends to smother the flame). There are several possible explanations for this difference, of which the most likely is that the temperature is sufficiently evenly distributed that soot is not formed and complete combustion occurs. Experiments by NASA reveal that diffusion flames in micro gravity allow more soot to be completely oxidized after they are produced than diffusion flames on Earth, because of a series of mechanisms that behave differently in micro gravity when compared to normal gravity conditions. These discoveries have potential applications in applied science and industry, especially concerning fuel efficiency.
In combustion engines, various steps are taken to eliminate a flame. The method depends mainly on whether the fuel is oil, wood, or a high-energy fuel such as jet fuel.
Flame temperatures
Temperatures of flames by appearance
It is true that objects at specific temperatures do radiate visible light. Objects whose surface is at a temperature above approximately 400
Watch movie Fire online on Amazon
Watch movie Fire online
Watch The Movie On PrimeDil Hai Ke Manta Nahin Full HD Movie Download

Chhoti Bahu Full HD Movie Download

The Power Man- Khalnayak Full HD Movie Download

Pyar Ka Sapna Full HD Movie Download

Devta Full HD Movie Download

Kabhi Socha Bhi Na Tha Full HD Movie Download

Aandhi (1975) Full HD Movie Download
.jpg)
Bazaar (1982) Full HD Movie Download
.jpg)
Kempegowda Full HD Movie Download

Carnival Full HD Movie Download

Phoenix The Warrior Full HD Movie Download

Datta Putrudu Full HD Movie Download

Swapna Lokam (1999) Full HD Movie Download
.jpg)
The Bucket List Full HD Movie Download

The Departed Full HD Movie Download

Manchal Nila Full HD Movie Download

Gali Medalu Full HD Movie Download

City War Full HD Movie Download

Anyay Abichar Full HD Movie Download

Lafangey Parindey Full HD Movie Download

Band Baaja Baaraat Full HD Movie Download

Download latest Movie from bollywood
- 1> baaghi 3
- 2> THE SKY IS PINK MOVIE FULL STORY AND REVIEW
- 3> Luka Chuppi
- 4> TO ALL THE BOYS I’VE LOVED BEFORE
- 5> Kabir Singh
- 6> Street Dancer 3D
- 7> Simmba
- 8> Gone Girl
- 9> The Girl Who Lived
- 10> Ludo
- 11> DILWALE DULHANIA LE JAYENGE
- 12> GUILTY
- 13> The Godfather
- 14> Adventures of Rusty
- 15> Sooryavanshi
- 16> Satyameva Jayate 2
- 17> Thappad
- 18> Bhool Bhulaiyaa 2
- 19> KGFChapter 2
- 20> Mardaani 2
- 21> Pinjar
- 22> Shivaji maharaj
- 23> Ek Villian 2
- 24> Hungama 2
- 25> Divergent
- 26> Mumbai Saga
- 27> The Internship
- 28> HIT (telugu)
- 29> Panga
- 30> The perfect date
- 31> 16 December
- 32> Gopala Gopala (Telugu)
- 33> Brahmastra
- 34> Gangubai Kathiawadi
- 35> Manmadhudu
- 36> Nenu local
- 37> Mahanati
- 38> Shatamanam bavathi
- 39> Lagaan
- 40> After
- 41> MOM
- 42> Shamshera
- 43> Raguvaran BTech
- 44> Khakee
- 45> The villain
- 46> OM
- 47> Mr. perfect
- 48> Bueatifull mind
- 49> Hichki
- 50> Gabbar Singh
- 51> Jogi
- 52> Before Sunrise
- 53> Before Sunset
- 54> Before Midnight
- 55> The Big Bull
- 56> Top Gun: Maverick
- 57> The Purge
- 58> The Sky is Pink
- 59> Laxmmi Bomb
- 60> Sadak 2
- 61> Sufna
- 62> Prithviraj
- 63> PK
- 64> Coolie No 1(2020)
- 65> Black Widow
- 66> Dear Zindagi
- 67> Dil Bechara
- 68> PHIR HERA PHERI
- 69> WAR
- 70> Dostana
- 71> RRR: Roudram Ranam Rudhiram
- 72> Maidan
- 73> Dabbang 3
- 74> Chhalaang
- 75> life as we know it
- 76> SherShaah
- 77> Sandeep Aur Pinky Faraar
- 78> Event Horizon
- 79> 83
- 80> Radhe: Your Most Wanted Bhai
- 81> Gunjan Saxena: The Kargil Girl
- 82> Mr India
- 83> Vivah
- 84> Anokha Bandhan
- 85> Ghost
- 86> Bhoot: Part One - The Haunted Ship
- 87> Haseen Dilruba
- 88> Laal Singh Chaddha
- 89> Qismat
- 90> Rajput
- 91> Drive
- 92> Dil Chahta Hai
- 93> Dil Ki Baazi
- 94> Dil Ka Rishta
- 95> Teesri Manzil
- 96> Dil
- 97> Love Aaj Kal
- 98> Khaali Peeli
- 99> Bunty Aur Babli 2
- 100> Atrangi Re
- 101> Gulabo Sitabo
- 102> Jodi
- 103> Suraj Pe Mangal Bhari
- 104> Deewana
- 105> Attack
- 106> Sardar Udham Singh
- 107> Toofan
- 108> THE LOVEBIRDS
- 109> Jersey
- 110> Ginny Weds Sunny
- 111> Thalaivi
- 112> Shiddat
- 113> Angels vs Zombies
- 114> Koi Mil Gya
- 115> Thank God
- 116> Bhuj: The Pride of India
- 117> Hum Aapke Hain Kaun
- 118> The Platform
- 119> Bird Box
- 120> Roohi Afzana
- 121> Torbaaz
- 122> Nikamma
- 123> World War Z
- 124> Extraction
- 125> Train to Busan
- 126> Life of Pi
- 127> SHAADI MEIN JROOR AANA
- 128> Himmat Aur Mehnat
- 129> To All The Boys: P.S. I Still Love You
- 130> Mimi
- 131> Good Newwz
- 132> Shubh Mangal Zyada Saavdhan
- 133> Raabta
- 134> Harry Potter and the Philosopher's Stone
- 135> Harry Potter and the Chamber of Secrets
- 136> Chhapaak
- 137> War of the Worlds
- 138> Harry Potter and the Prisoner of Azkaban
- 139> Harry Potter and the Goblet of Fire
- 140> MURDER MYSTERY
- 141> Shakuntala Devi
- 142> Bachchan Pandey
- 143> Jayeshbhai Jordar
- 144> Sheer Qorma
- 145> Saina
- 146> 'O' Pushpa I hate tears
- 147> Kedarnath
- 148> MS Dhoni The Untold Story
- 149> Chhichhore
- 150> Badhaai Ho
- 151> Unstoppable
- 152> Oz the Great And Powerful
- 153> The Girl on the Train
- 154> Haathi Mere Saathi 2020
- 155> The Conjuring: The Devil Made Me Do It
- 156> Gandhi Se Pehle Gandhi
- 157> The Song of Scorpions
- 158> Srimanthudu
- 159> Hello Guru Prema Kosame
- 160> Beauty and The Beast
- 161> Black Panther
- 162> Charlie and the Chocolate Factory
- 163> Bole Chudiyan
- 164> Fidaa
- 165> Duvvada Jagannadham
- 166> Bruce Lee: The Fighter
- 167> Hyper
- 168> Yaara
- 169> Red (2020)
- 170> Shivam
- 171> That Is Mahalakshmi
- 172> Nishabdham
- 173> Aashram 2020 web series
- 174> Laxmii
- 175> Mismatched
- 176> STUDENT OF THE YEAR 2
- 177> NAIL POLISH
- 178> Ramprasad Ki Tehrvi
- 179> KAAGAZ
- 180> 12 o Clock
- 181> The Power
- 182> bolo hau
- 183> Tribhanga
- 184> JAMUN
- 185> Madam Chief Minister
- 186> Maasaab
- 187> Aadhaar
- 188> Tanhaji
- 189> Bhaagi 3
- 190> Bhootnath
- 191> MALANG
- 192> Jai Mummy Di
- 193> Haathi Mere Saathi 2021
- 194> Shakeela
- 195> Unpaused
- 196> Annayya
- 197> Vamsoddharakudu
- 198> Mrugaraju
- 199> Narasimha Naidu
- 200> Sankranti
- 201> Manasu Maata Vinadhu
- 202> Anjaane
- 203> Apaharan
- 204> Bachke Rehna Re Baba
- 205> Bewafaa
- 206> Roohi
- 207> Radhe
- 208> Zindagi Khoobsoorat Hai
- 209> Yeh Mohabbat Hai
- 210> Yeh Kya Ho Raha Hai?
- 211> The Tomorrow War
- 212> DehradunDiary
- 213> Meri Shaadi Karaoo
- 214> Matruu Ki Bijlee Ka Mandola
- 215> No One Killed Jesica
- 216> Aag Ka Goola
- 217> Eight Million Dollars
- 218> Three Hundred
- 219> Cats and Dog
- 220> Decoy
- 221> Gold Rush
- 222> You Have Got Mail
- 223> Final Destination three
- 224> Tofan
- 225> Jungle
